ASND Breakout Report
Investor Analysis
ASND: Investor Summary (Ascendis Pharma A/S)
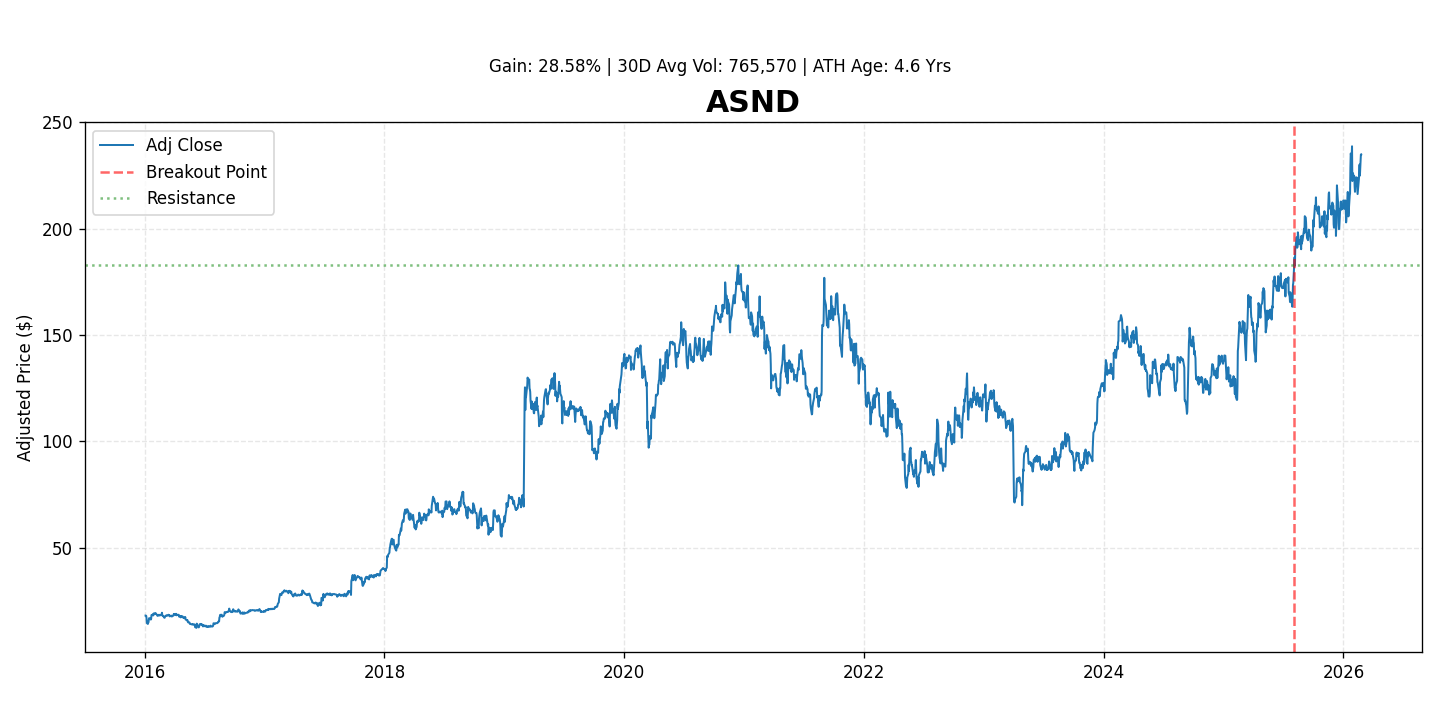
Ascendis Pharma A/S (NASDAQ: ASND) is a dynamic biopharmaceutical company leveraging its proprietary TransCon™ technology platform to develop innovative prodrugs for rare diseases and oncology. The company has demonstrated strong momentum, with its stock gaining 28.6% since its recent breakout, driven by key pipeline advancements and commercial progress.
Business Overview
Ascendis Pharma is focused on applying its TransCon™ (Transient Conjugation) technology to create best-in-class therapies that offer sustained and predictable drug exposure, improving efficacy, safety, and patient convenience. Its pipeline targets critical unmet needs in endocrinology and oncology.
- Commercial Product: Skytrofa® (TransCon™ hGH) for pediatric growth hormone deficiency, approved in the US, EU, and Japan, demonstrating robust commercial uptake.
- Lead Pipeline Product: TransCon™ PTH (Palopegteriparatide / Yorvigo®) for hypoparathyroidism, approved in the EU and awaiting potential US approval after a resubmitted New Drug Application (NDA). This represents a significant potential first-in-class therapy for chronic hypoparathyroidism.
- Advanced Pipeline: TransCon™ CNP for achondroplasia and TransCon™ Trastuzumab ADC for HER2+ solid tumors are in clinical development.
Key Competitive Moats
- Proprietary TransCon™ Technology: This innovative platform allows for the creation of sustained-release prodrugs, offering a differentiated profile over existing treatments by providing consistent therapeutic levels and reducing dosing frequency.
- First-in-Class Potential: TransCon™ PTH (Yorvigo®) is poised to be a groundbreaking treatment for chronic hypoparathyroidism, a rare condition with no approved hormone replacement therapy in the US.
- Validated Platform: The successful approval and commercialization of Skytrofa® validate the TransCon™ platform's ability to deliver effective and approvable therapies.
- Diversified Pipeline: A robust pipeline beyond Skytrofa and TransCon PTH, addressing multiple rare diseases and oncology, reduces single-product dependency and offers multiple shots on goal.
Revenue and Earnings Growth Projections (2025-2026)
Ascendis Pharma is projected to enter a period of accelerated revenue growth, primarily driven by the anticipated US commercial launch of TransCon PTH (Yorvigo®) and continued uptake of Skytrofa®. While the company is currently investing heavily in R&D and commercial infrastructure, leading to projected net losses, these are expected to narrow significantly.
- Fiscal Year 2025 (ending December 31, 2025): Analyst consensus estimates forecast revenues in the range of $450-$500 million, a substantial increase from 2024, reflecting the anticipated strong launch trajectory of Yorvigo.
- Fiscal Year 2026 (ending December 31, 2026): Revenue projections further climb to an estimated $650-$750 million, indicating strong year-over-year expansion as Yorvigo gains market traction and Skytrofa maintains growth.
Losses per share are expected to narrow considerably across these years, with a potential pathway to positive EPS emerging by late 2026 or early 2027, contingent on successful regulatory and commercial execution for Yorvigo.
Recent Catalysts
- TransCon PTH US PDUFA Date: The critical PDUFA action date for the resubmitted NDA for TransCon PTH (Yorvigo®) in the US is **May 14, 2024**, with a positive decision expected.
- Potential US Commercial Launch of Yorvigo: A swift launch post-approval in H2 2024 would unlock a significant revenue stream.
- Continued Skytrofa® Uptake: Strong Q1 2024 and ongoing sales growth for Skytrofa® in existing and new markets.
- Pipeline Advancements: Further clinical data readouts from TransCon CNP and TransCon Trastuzumab ADC could provide additional long-term value drivers.
Main Risks
- Regulatory Risk: While approval for TransCon PTH is anticipated, regulatory decisions are never guaranteed, and any further delays or a Complete Response Letter (CRL) would negatively impact the stock.
- Commercialization Challenges: Successful market penetration and reimbursement for Yorvigo® will be crucial. Competition and payer access issues could limit upside.
- Clinical Trial Risk: Future pipeline candidates (e.g., TransCon CNP, TransCon Trastuzumab ADC) may not achieve desired clinical endpoints or regulatory approval.
- Competition: While Yorvigo offers a novel approach, other companies may develop competing therapies or existing palliative treatments could remain entrenched.
- Cash Burn and Funding: As a biotech company in growth phase, Ascendis has a high cash burn rate. Future capital raises, potentially dilutive, cannot be ruled out.