CELC Breakout Report
Investor Analysis
Investor Summary: CELC (Celcuity Inc.)
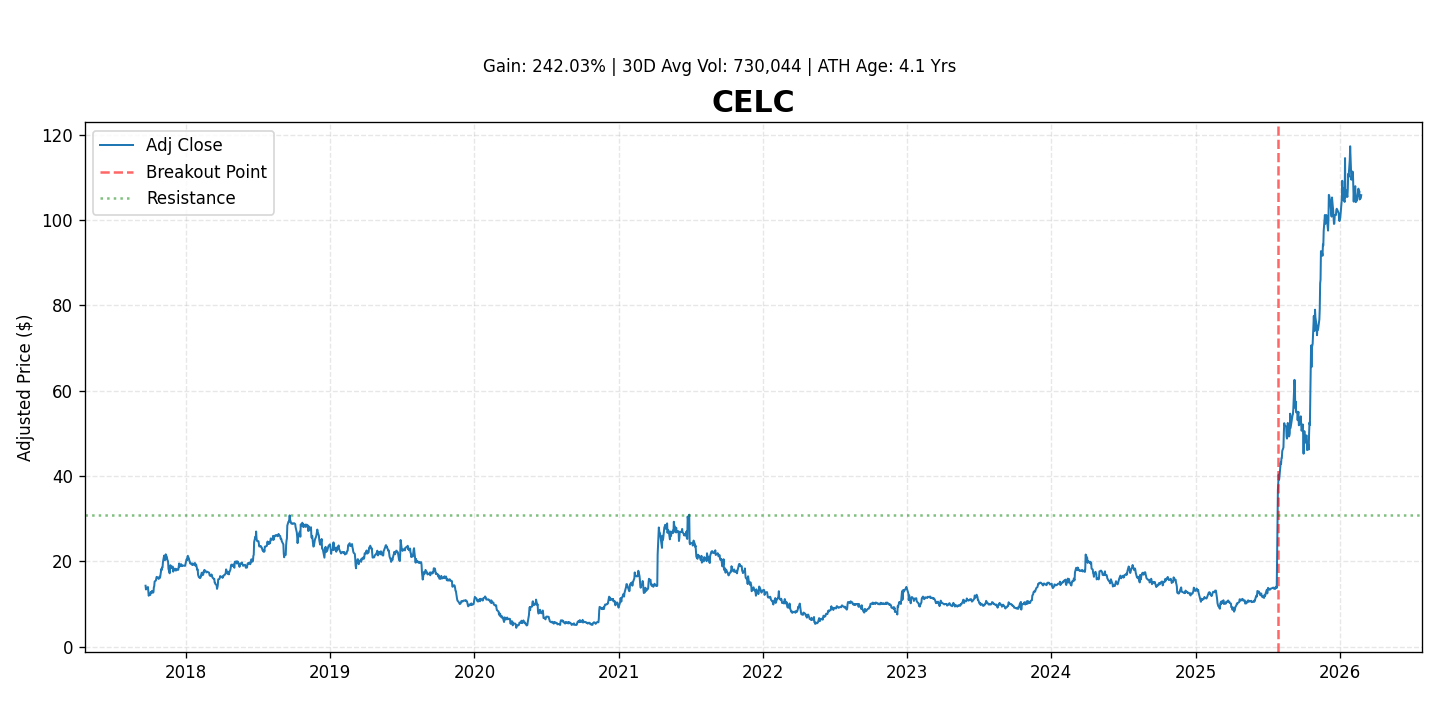
Celcuity Inc. (NASDAQ: CELC) is a clinical-stage biotechnology company developing innovative diagnostic tests to improve outcomes for cancer patients. The company’s proprietary CELx platform aims to identify the specific cell signaling abnormalities driving an individual's cancer, enabling oncologists to select the most effective targeted therapies and predict drug response. Following a significant breakout, the stock has gained 242.0%, reflecting increased investor optimism in its platform and commercialization prospects.
Business Overview
Celcuity is focused on precision medicine in oncology, with its lead product, the CELx HRD test, designed to identify patients with homologous recombination deficiency (HRD) who are most likely to benefit from PARP inhibitors. Beyond HRD, the company's robust pipeline leverages its functional profiling technology across various cancer types (e.g., HR+/HER2- breast cancer) to develop companion diagnostics and predict therapeutic efficacy. This technology evaluates drug-induced changes in live tumor cells, offering a unique approach complementary to traditional genomic sequencing.
Key Competitive Moats
- Proprietary CELx Platform: A differentiated functional profiling technology that analyzes drug response directly on living tumor cells, providing insights beyond genomic mutations.
- Strong Clinical Validation: The platform has demonstrated significant clinical utility and predictive power in various studies, including the D-DREAM trial, building a robust evidence base.
- Intellectual Property: Extensive patent portfolio protecting its CELx technology, methods, and specific diagnostic applications.
- Strategic Partnerships: Collaborations with leading pharmaceutical companies for companion diagnostic development and commercialization, providing critical funding and market access.
- First-Mover Advantage: A unique approach in functional diagnostics for specific cancer indications positions Celcuity as a pioneer in certain segments of precision oncology.
Revenue and Earnings Growth Projections (2025/2026)
As an early-stage commercial biotech, Celcuity is focused on market penetration and pipeline development. While specific quarterly guidance is not yet provided by the company, analyst consensus and expected commercial ramp-up suggest significant revenue acceleration, with continued investment in R&D and commercial infrastructure resulting in projected losses through 2025 and potential profitability towards late 2026.
- Q1 2025 (Jan-Mar): Revenue: $2.5M - $3.5M | EPS: -$0.28 to -$0.22 (Loss)
- Q2 2025 (Apr-Jun): Revenue: $3.5M - $5.0M | EPS: -$0.25 to -$0.18 (Loss)
- Q3 2025 (Jul-Sep): Revenue: $5.0M - $7.0M | EPS: -$0.22 to -$0.15 (Loss)
- Q4 2025 (Oct-Dec): Revenue: $7.0M - $9.0M | EPS: -$0.20 to -$0.12 (Loss)
- Q1 2026 (Jan-Mar): Revenue: $9.0M - $12.0M | EPS: -$0.15 to -$0.08 (Loss)
- Q2 2026 (Apr-Jun): Revenue: $12.0M - $16.0M | EPS: -$0.10 to -$0.02 (Loss)
- Q3 2026 (Jul-Sep): Revenue: $16.0M - $22.0M | EPS: Break-even to $0.05 (Potential Profitability)
- Q4 2026 (Oct-Dec): Revenue: $22.0M - $30.0M | EPS: $0.05 to $0.15 (Profit)
Recent Catalysts
- Positive Clinical Data: Continued release of encouraging data from trials like D-DREAM, reinforcing the clinical utility and predictive power of the CELx platform.
- Commercial Expansion: Ongoing rollout and increasing adoption of the CELx HRD test by oncology practices.
- New Pharmaceutical Partnerships: Formation of additional collaborations for companion diagnostic development, expanding pipeline and revenue opportunities.
- Regulatory Milestones: Progress towards FDA approvals or clearances for new diagnostic tests or companion diagnostics.
- Presentations at Major Medical Conferences: Highlighting research and clinical findings to a broad audience of oncologists and researchers.
Main Risks
- Clinical Trial Risk: Failure of ongoing or future clinical trials to demonstrate sufficient utility or efficacy of their diagnostic tests.
- Regulatory Approval Risk: Delays or failure to secure necessary regulatory approvals (e.g., FDA clearance) for new tests.
- Market Adoption & Reimbursement: Challenges in achieving widespread adoption by oncologists and securing adequate reimbursement from payers.
- Competition: Intense competition from established diagnostic companies and other emerging precision medicine technologies.
- Cash Burn & Funding: As an early-stage company, CELC has a significant cash burn rate and may require additional capital raises, potentially diluting existing shareholders.
- Intellectual Property Disputes: Potential for challenges to its patent portfolio or infringement claims from competitors.
- Dependence on Partnerships: Success is highly dependent on the performance and continued collaboration with pharmaceutical partners.
Disclaimer: This summary is for informational purposes only and does not constitute financial advice. Investment in early-stage biotechnology companies carries significant risks, and actual results may differ materially from projections. Investors should conduct their own due diligence and consult with a qualified financial advisor.
```