TRVI Breakout Report
Investor Analysis
Investor Summary: Trevi Therapeutics (TRVI)
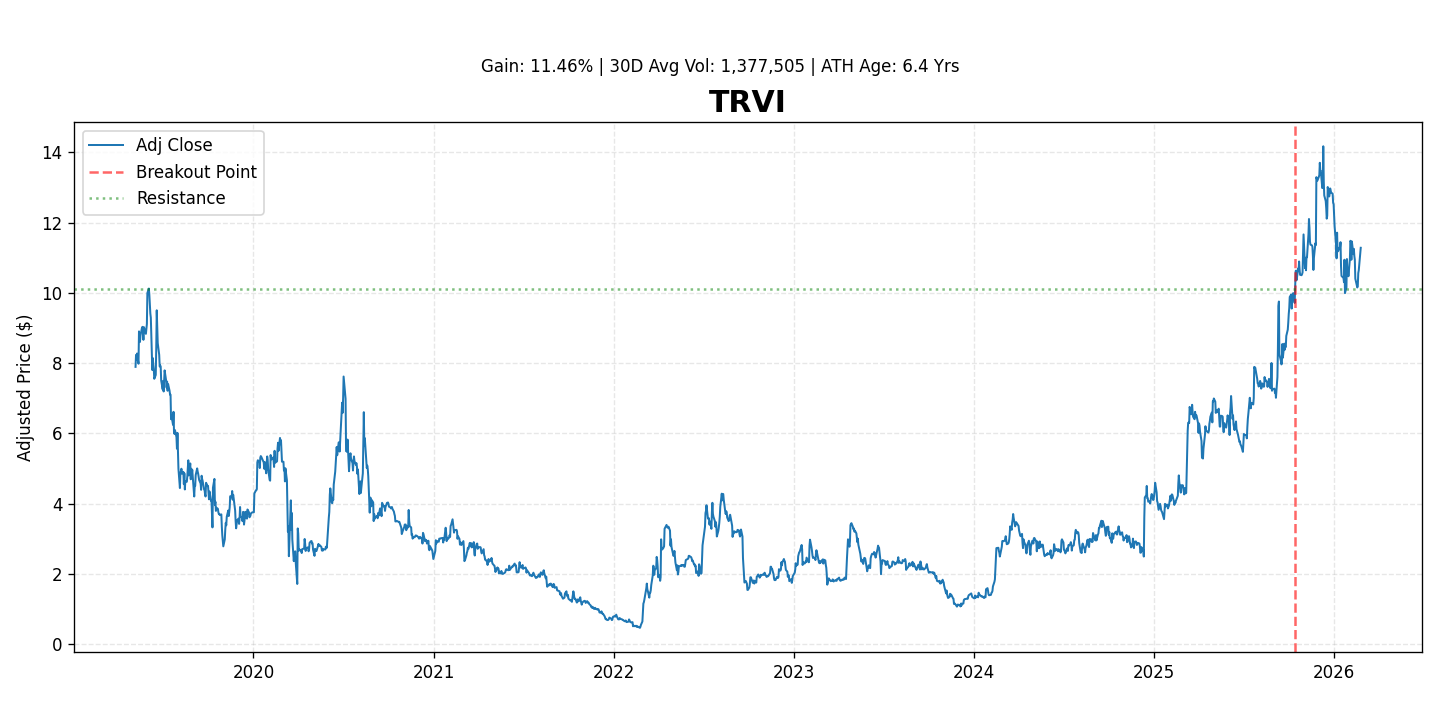
Trevi Therapeutics (NASDAQ: TRVI) is a clinical-stage biopharmaceutical company focused on developing Haduvio (nalbuphine ER) for the treatment of chronic neurological conditions. The stock has recently demonstrated positive momentum, gaining 11.5% since its breakout, reflecting growing investor interest and confidence in its clinical pipeline.
Business Overview
Trevi Therapeutics is dedicated to improving the lives of patients suffering from chronic neurological conditions. Its lead product candidate, Haduvio (nalbuphine ER), is an oral, extended-release formulation of nalbuphine, a dual µ-opioid receptor antagonist and κ-opioid receptor agonist. Haduvio is currently being developed for two primary indications:
- Prurigo Nodularis (PN): A chronic, debilitating skin disease characterized by intense, persistent itch and the formation of numerous skin lesions. Haduvio is in a pivotal Phase 2b/3 trial (PRISM trial).
- Chronic Cough in Idiopathic Pulmonary Fibrosis (IPF): A severe and persistent cough that significantly impacts the quality of life for patients with IPF. Haduvio is in a Phase 2 trial (CANAL trial).
Key Competitive Moats
- Unique Mechanism of Action: Haduvio’s dual opioid receptor modulation offers a distinct therapeutic approach for managing chronic itch and cough, potentially superior to single-target therapies.
- Orphan Drug Designation: Haduvio has received Orphan Drug Designation from both the U.S. FDA and the European Medicines Agency (EMA) for Prurigo Nodularis, which provides incentives such as market exclusivity upon approval and development benefits.
- Pivotal Trial Stage: The PRISM trial for PN is a Phase 2b/3 study, positioning Haduvio closer to potential regulatory approval and commercialization compared to earlier-stage assets.
- Addressing Unmet Needs: Both Prurigo Nodularis and chronic IPF cough represent areas with significant unmet medical needs and limited approved treatment options, offering a substantial market opportunity.
Revenue and Earnings Growth (2025 and 2026)
As a clinical-stage biopharmaceutical company, Trevi Therapeutics is not yet generating significant product revenue. The company is currently investing heavily in research and development for Haduvio. Significant product revenue is not anticipated in 2025 and is unlikely in early to mid-2026, as Haduvio is still in clinical trials and awaiting potential regulatory approvals. The company is expected to continue reporting operating losses through this period as it advances its clinical programs and prepares for potential commercialization.
- Q1 2025 (Expected): Continued R&D expenses, net loss anticipated.
- Q2 2025 (Expected): Continued R&D expenses, potential for NDA filing for PN if PRISM data is positive, net loss anticipated.
- Q3 2025 (Expected): Continued R&D expenses, ongoing regulatory review if NDA filed, net loss anticipated.
- Q4 2025 (Expected): Continued R&D expenses, ongoing regulatory review, net loss anticipated.
- Q1 2026 (Expected): Continued R&D/pre-commercial expenses, potential for PDUFA date if NDA filed in late 2025, net loss anticipated.
- Q2 2026 (Expected): Continued R&D/pre-commercial expenses, potential for initial commercial launch if approved, net loss anticipated.
Recent Catalysts
- Anticipated Top-line Data for PRISM Trial: Top-line data from the pivotal Phase 2b/3 PRISM trial for prurigo nodularis is expected in the first half of 2025, a critical de-risking event.
- Interim Analysis for CANAL Trial: An interim analysis for the Phase 2 CANAL trial in chronic cough in IPF is anticipated in Q3 2024, providing an early look at efficacy and safety.
- Strong Cash Position: The company reported a cash runway into Q4 2025 (as of Q1 2024), providing financial stability to execute on key milestones.
- Positive Market Sentiment: The recent 11.5% stock gain reflects positive investor sentiment, possibly driven by progress in trials, anticipation of data readouts, or broader interest in the neurological rare disease space.
Main Risks
- Clinical Trial Risk: Haduvio may fail to meet primary or secondary endpoints in ongoing or future clinical trials (PRISM or CANAL), leading to delays or discontinuation of development.
- Regulatory Risk: Even with positive data, there is no guarantee of regulatory approval from the FDA or other agencies. The regulatory pathway can be complex and unpredictable.
- Competition: The markets for prurigo nodularis and chronic cough are evolving, with other companies developing treatments. Future competition could impact market share and pricing.
- Financing Risk: As a pre-revenue company, Trevi relies on capital raises. Future financing rounds may be necessary, potentially leading to shareholder dilution.
- Commercialization Risk: If approved, successful commercialization depends on market access, physician adoption, and reimbursement, which can be challenging for new therapies.
Disclaimer: This summary is for informational purposes only and does not constitute financial advice. Investors should conduct their own due diligence and consult with a financial professional before making any investment decisions. Projections are forward-looking and subject to change.
```